Model parameters were estimated by maximum likelihood using nonlinear regression (NONMEM, v. 
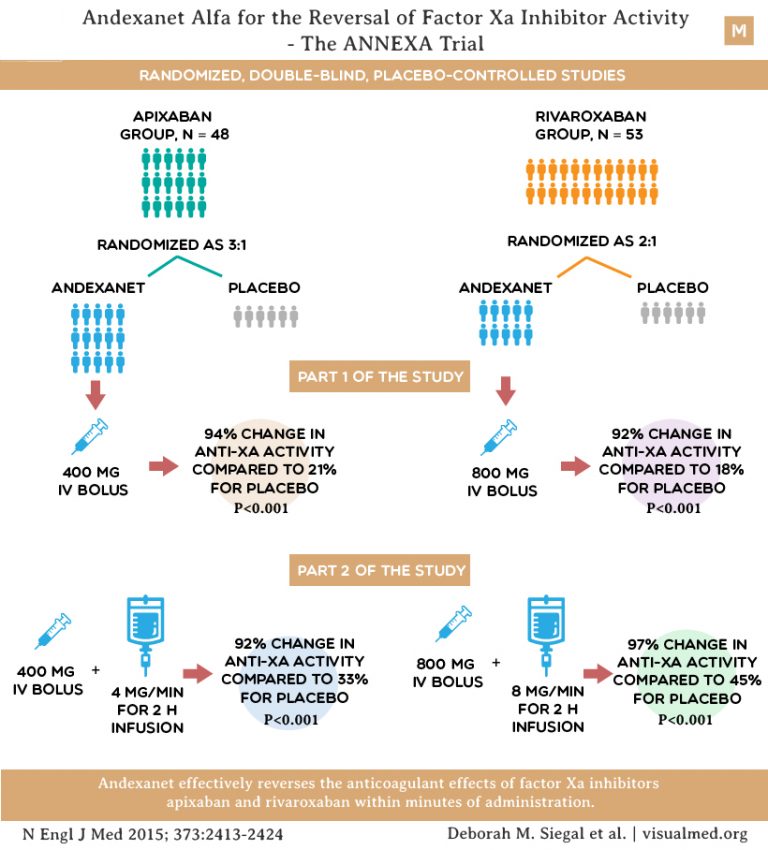
We report a PKPD model that accounts for this extravascular-intravascular redistribution and allows determination of the appropriate AnXa dose to reverse the anti-fXa activity for each approved direct fXa inhibitor.Ī model for rivaroxaban was constructed first by jointly analyzing AnXa concentrations, total and unbound rivaroxaban concentrations, and anti-fXa activity for 5 different doses of AnXa (each administered at the peak plasma level of 20 mg QD rivaroxaban at steady state). Reversal of fXa inhibitor-induced anticoagulation requires a molar excess of AnXa relative to the total amount of fXa inhibitor in the blood – i.e., the initial fXa inhibitor plasma concentration plus the redistributed amount. The decreased unbound concentrations of the direct fXa inhibitors (apixaban, rivaroxaban and edoxaban) cause a portion of the extravascular fXa inhibitors to move into the vasculature. AnXa binds fXa inhibitors, sequestering them in plasma, thereby reducing the free inhibitor concentration, reversing fXa inhibition, and normalizing thrombin generation. AnXa is a recombinant, engineered version of human fXa that is catalytically inactive but retains high affinity for direct fXa inhibitors. Andexanet alfa (AnXa) is a specific antidote under development for direct and indirect factor Xa (fXa) inhibitors. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
